Chemical EquilibriumHard
Question
For the equilibrium CuSO4.5H2O(s) ⇋ CuSO4.3H2O(s) + 2H2O(g)
Kp = 2.25 × 10-4 atm2 and vapour pressure of water is 22.8 Torr at 298 K.
CuSO4.5H2O(s) is efflorescent (i.e., loses water) when relative humidity is :
Kp = 2.25 × 10-4 atm2 and vapour pressure of water is 22.8 Torr at 298 K.
CuSO4.5H2O(s) is efflorescent (i.e., loses water) when relative humidity is :
Options
A.less than 33.3%
B.less than 50 %
C.less than 66.6%
D.above 66.6%
Solution
CuSO4 . 5H2O(s) ⇋ CuSO4 . 3H2O(s) + 2H2O(g) KP = 2.25 × 10-4
KP = P2H2O = 2.25 × 10-4 PH2O = 1.5 × 10-2
Vapour Pr =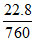 = 3 × 10-2 R.H. =
= 3 × 10-2 R.H. = 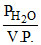 × 100 = 50%
× 100 = 50%
Therefore, (B) option is correct.
KP = P2H2O = 2.25 × 10-4 PH2O = 1.5 × 10-2
Vapour Pr =
Therefore, (B) option is correct.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
If the value of an equilibrium constant for a particular reaction is 1.6 × 1012, then at equilibrium the system wil...The dissociation of ammonium carbamate may be represented by the equation NH4CO2NH2(s) ⇋ 2NH3(g) + CO2(g)ᐃH0...Introduction of inert gas (at the same temperature) will affect the equilibrium if :...If for 2A2B(g) ⇋ 2A2(g) + B2(g), Kp = TOTAL PRESSURE (at equilibrium) and starting the dissociation from 4 mol of ...In which of the following equilibrium Kc and Kp are not equal ?...