Chemical BondingHard
Question
If a molecule MX3 has zero dipole moment, the sigma bonding orbitals used by M (atomic number < 21) are :
Options
A.pure p
B.sp hybridised
C.sp2 hybridised
D.sp3 hybridised
Solution
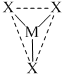 , sp2 - hybridised orbitals will be arranged in planar triangle manner and μ = 0
, sp2 - hybridised orbitals will be arranged in planar triangle manner and μ = 0Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Lattice energy of an ionic compounds depends upon...Treatment of cyclopentanone with methyl lithium gives which of the following species?...Match list-I with list-II and select List-I List-II(A) Zeiglernata catalyst (i) Fe4[Fe(CN)6]3(B) Brown ring complex (ii)...Incorrect order according to the property is :...Under hydrolytic conditions, the compounds used for preparation of linear polymer and for chain termination, respectivel...