Chemical EquilibriumHard
Question
For the reaction : PCl5 (g) ⇋ PCl3 (g) + Cl2 (g)
The forward reaction at constant temperature is favoured by
The forward reaction at constant temperature is favoured by
Options
A.introducing chlorine gas at constant volume
B.introducing an inert gas at constant pressure
C.increasing the volume of the container
D.introducing PCl5 at constant volume
Solution
According to KP = 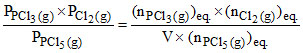
and on adding inert gas at constant pressure effect on equilibrium will be similar to as if volume of container has been increased.
and on adding inert gas at constant pressure effect on equilibrium will be similar to as if volume of container has been increased.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
For the reaction 2NOCl(g)$\rightleftharpoons$ 2NO(g) + Cl2(g), ΔHo = 18 kcal and ΔSo = 30 cal/K at 300 K. The equilibriu...For the reaction CaCO3(s) ⇋ CaO(s) + CO2(g), the pressure of CO2(g) depends on :...Densities of diamond and graphite are 3.5 and 2.3 g/mL. C (diamond) ⇋ C (graphite) ᐃrH = - 1.9 kJ/molefavour...In a reaction carried out at 500 K, 0.001% of the total number of collisions are effective. The energy of activation of ...The equilibrium constant for the reaction N2(g) + O2(g) $\rightleftharpoons$ 2NO(g) is K1 and the equilibrium constant f...