Chemical EquilibriumHard
Question
In a reversible reaction A 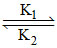 B, the initial concentration of A and B are a and b in moles per litre and the equilibrium concentrations are ( a - x) and (b + x) respectively; express x in terms of k1, k2, a and b.
B, the initial concentration of A and B are a and b in moles per litre and the equilibrium concentrations are ( a - x) and (b + x) respectively; express x in terms of k1, k2, a and b.
Options
A.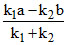
B.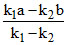
C.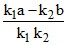
D.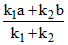
Solution
A 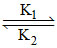 B Kc =
B Kc = 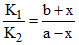
a - x b + x x =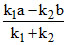 Therefore, (A) option is correct.
Therefore, (A) option is correct.
a - x b + x x =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Which of the following is/are correct statement(s)?...The amounts of 0.8 mol of PCl5 and 0.2 mole of PCl3 are mixed in a 1 L flask. At equilibrium, 0.4 mole of PCl3 is presen...At 525 K, PCl5(g) is 80% dissociated at a pressure of 1 atm. Now, sufficient quantity of an inert gas at constant pressu...For the reaction $$\text{CO}(g) + \text{H}_2\text{O}(g) \rightleftharpoons \text{CO}_2(g) + \text{H}_2(g)$$ at a given t...An amount of 1 mole of PCl3(g) and 1 mole of PCl5(g) is taken in a vessel of 10 L capacity maintained at 400 K. At equil...