Atomic StructureHard
Question
Which of the following statement (s) is (are) correct ?
Options
A.The electroni configuration of Cr is [Ar] 3d54s1 (Atomic number of Cr = 24)
B.The magnetic quantm number may have a negative value
C.In silver atom 23 electrons have a spin of one type and 24 of the opposite type (Atomic number of Ag = 27)
D.The oxidation state of nitrogen in HN3 is
Solution
(a) 24Cr = [Ar]3d5, 4s1
(b) m = - l to + l including zero.
(c) 47Ag = 1s2, 2s2, 2p6 3s2, 3p6, 3d10, 4s2, 4sp6, 4d10, 5s1
Thus 24 electron have s = + while rest 23 have. s = -
while rest 23 have. s = - 
(d) Oxidation number of N in NH3 is -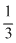
(b) m = - l to + l including zero.
(c) 47Ag = 1s2, 2s2, 2p6 3s2, 3p6, 3d10, 4s2, 4sp6, 4d10, 5s1
Thus 24 electron have s = +
 while rest 23 have. s = -
while rest 23 have. s = - 
(d) Oxidation number of N in NH3 is -
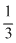
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The quantum number which determines the shape of the orbital is...If there are three possible values (–1/2, 0, +1/2) for the spin quantum number, then the maximum capacity of second orbi...The element having no neutron in the nucleus of its atom is...The ratio of the speed of the electron in the ground state of hydrogen atom to the speed of light in vacuum is...Photons of equal energy were incident on two different gas samples. One sample containing H-atoms in the ground state an...