SolutionHard
Question
Two beakers, one containing 20 ml of a 0.05 M aqueous solution of a non volatile, non electrolyte and the other, the same volume of 0.03 M aqueous solution of NaCl, are placed side by side in a closed enclose. What are the volumes in the two beakers when equation is attached ? Volume of the solution in the first and second beaker are respectively
Options
A.21.8 ml and 18.2 mL
B.18.2 mL and 21.8 mL
C.20 mL and 20 mL
D.17.1 mL and 22.9 mL
Solution
Mole of solute in first beaker = 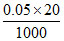 = 0.001
= 0.001
mole of solute (Na+ & Cl-) in other beaker =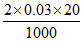 = 0.0012
= 0.0012
conc. of IInd beaker is higher then Ist beaker so water flowes from Ist beaker to IInd beaker till both beaker achieved equal conc. let v volume of water flows from Ist to IInd beaker
so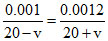
v = 1.8 ml
volume of Ist beaker = 20 - 1.8 = 18.2 ml
volume of IInd beaker = 20 + 1.8 = 21.8 ml.
mole of solute (Na+ & Cl-) in other beaker =
conc. of IInd beaker is higher then Ist beaker so water flowes from Ist beaker to IInd beaker till both beaker achieved equal conc. let v volume of water flows from Ist to IInd beaker
so
v = 1.8 ml
volume of Ist beaker = 20 - 1.8 = 18.2 ml
volume of IInd beaker = 20 + 1.8 = 21.8 ml.
Create a free account to view solution
View Solution FreeMore Solution Questions
A solution when diluted with H2O and boiled, it gives a white precipitate. On addition of excess NH4Cl/NH4OH. the volume...A vessel has nitrogen gas and water vapours in equilibrium with liquid water at a total pressure of 1 atm. The partial p...Aqueous solutions of Na2S2O3 on reaction with Cl2 gives...Consider the following reactions.(Hot solution)In the above reactions, and X are respectively....In a 0.2 molal aqueous solution of a weak acid HX the degree of ionization is 0.3. Taking kf for water as 1.85, the free...