SolutionHard
Question
A solute′S′ undergoes a reversible trimerization when dissolved in a certain solvent. The boiling point elevation of its 0.1 molal solution was found to be identical to the boiling point elevation in case of a 0.08 molal solution of a solute which neither undergoes association nor dissociation. To what percent had the solute ′S′ undergone trimerization?
Options
A.30%
B.40%
C.50%
D.60%
Solution
3S ⇋ S3
1-α α/3 ⇒ i = 1 -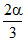
Now 0.1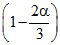 = 0.08
= 0.08
⇒ α = 0.3. Hence 30% trimerization.
1-α α/3 ⇒ i = 1 -
Now 0.1
⇒ α = 0.3. Hence 30% trimerization.
Create a free account to view solution
View Solution FreeMore Solution Questions
An aqueous solution of gas (X) gives the white turbidity on passing H2S in the solution. Identify (X)...A liquid solution is formed by mixing 10 moles of aniline and 20 moles of phenol at a temperature where the vapour press...The limiting value of van’t Hoff factor for Na2SO4 is...1 mol each of 1the following compounds is dissolved in 1L of solution. Which will have the largest ᐃTb value ?...A current of 2.6 ampere was passed through CuSO4 solution for 380 sec. The amount of Cu deposited is (at. wt. of Cu = 63...