SolutionHard
Question
Consider two liquids A & B having pure vapour pressures PAo & PBo forming an ideal solution. The plot of 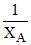 v/s
v/s 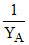 (where XA and YA are the mole fraction of liquid A in liquid and vapour phase respectively) is linear with slope and Y intercepts respectively :
(where XA and YA are the mole fraction of liquid A in liquid and vapour phase respectively) is linear with slope and Y intercepts respectively :
Options
A.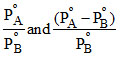
B.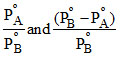
C.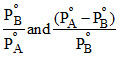
D.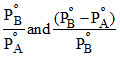
Solution
P′A = PAo XA and P′B = PoB XB
P′A = PM . YA and P′B = PM .YB
∴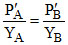
or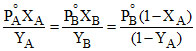 or
or 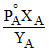 (1 - YA) = PoB - PoB XA
(1 - YA) = PoB - PoB XA
or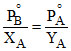 + (PoB - PoA) or
+ (PoB - PoA) or 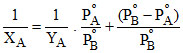
or y = mx + C ∴ Slope = m =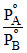 and intercept C =
and intercept C = 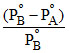
P′A = PM . YA and P′B = PM .YB
∴
or
or
or y = mx + C ∴ Slope = m =
Create a free account to view solution
View Solution FreeMore Solution Questions
A binary liquid solution is prepared by mixing n-heptane and ethanol. Which one of the following statements is correct r...Pure water boils at 373 K and nitric acid at 359 K. The azeotropic mixture of water and nitric acid boils at 393.5 K. On...Relative decrease in vapour pressure of an aqueous solution containing 2 moles [Cu(NH3)3Cl] Cl in 3 moles H2O is 0.50. O...When an ideal binary solution is in equilibrium with its vapour, molar ratio of the two components in the solution and i...Of the following measurements, the one most suitable for the determination of the molecular mass of oxyhaemoglobin, a mo...