SolutionHard
Question
The solubility of N2(g) in water exposed to the atmosphere, when the partial pressure is 593 mm is
5.3 × 10-4 M. Its solubility at 760 mm and at the same temperature is :
5.3 × 10-4 M. Its solubility at 760 mm and at the same temperature is :
Options
A.4.1 × 10-4 M
B.6.8 × 10-4 M
C.1500 M
D.2400 M
Solution
Solubility ∝ pressure
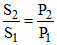
S2 = 5.3 × 10-4 ×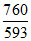 = 6.8 × 10-4 M
= 6.8 × 10-4 M
S2 = 5.3 × 10-4 ×
Create a free account to view solution
View Solution FreeMore Solution Questions
A very small amount of non-volatile solute (that does not dissociate) is dissolved in 56.8 cm3 of benzene (density 0.889...The degree of dissociation (α) of a weak electrolyte, Ax By is related to van′t Hoff factor (i) by the expres...Among the following substances, the lowest vapour pressure is exerted by-...At higher altitudes, water boils at temperature < 100oC because...The concentration of pollutant in ppm (w/w), that has been measured at 450 mg per 150 kg of sample is :...