Nitrogen Containing CompoundHard
Question
Examine the following two structures for the anilinium ion and choose the corect statement from the ones given below :
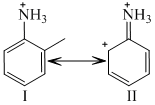

Options
A.II is not an acceptable canonical structure because carbonium ions are les stable than ammonium ions
B.II is not an acceptable canonical structure because it is non-aromatic
C.II is not an acceptable canonical structure because the nitrogen has 10 valence electrons
D.II is an acceptable canonical structure
Solution
In structure II, N-atom
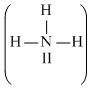 has 10 electrons which is impossible, thus the structure II is not acceptable.
has 10 electrons which is impossible, thus the structure II is not acceptable.
 has 10 electrons which is impossible, thus the structure II is not acceptable.
has 10 electrons which is impossible, thus the structure II is not acceptable.Create a free account to view solution
View Solution FreeMore Nitrogen Containing Compound Questions
Which of the following will be most stable diazonium salt RN2+X- ?...A compound contains 69.5% oxygen and 30.5% nitrogen and its molecular weight is 92. The formula of the compound is -...Nitrogen is liberated by the thermal decomposition of only :-...Which of the following gives primary amine on reduction ?...In Duma′s method for estimation of nitrogen, 0.25 g of an organic compound gave 40 mL of nitrogen collected at 300...