Coordination CompoundHard
Question
For the reaction, Ni2+ + 4NH3 ⇋ [Ni(NH3)4]2+ at equilibrium, if the solution contains 1.6 ×10-4% of nickel in the free state, and the concentration of NH3 at equilibrium is 0.5 M. Then the instability constant of the complex will be approximately equal to :
Options
A.1.0 × 10-5
B.1.5 × 10-16
C.1.0 × 10-7
D.1.5 × 10-17
Solution
Ni2+ + 4 NH3 ⇋ [Ni(NH3)4]2+
∴ k =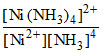
But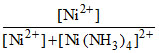 = 1.6 × 10-6 or
= 1.6 × 10-6 or 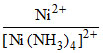 ≈ 1.6 × 10-6
≈ 1.6 × 10-6
∴ k =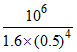 = 107
= 107
Hence instability constant = 10-7
∴ k =
But
∴ k =
Hence instability constant = 10-7
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Select the correct statement among the following:...Cis-trans isomerism is found in square planar complexes of molecular formula (′a′ and ′b′ are mo...Glucose and Fructose are :-...The number of sigma bonds in Zeise′s salt is:...The complex [Pt(NH3)4]2+ has ..... structure :...