Coordination CompoundHard
Question
All the metal ions contains t2g 6eg0 configurations. Which of the following complex will be paramagnetic?
Options
A. [FeCl(CN)4(O2)]4-
B.K4[Fe(CN)6]
C.[Co(NH3)6]Cl3
D.[Fe(CN)5(O2)]-5
Solution
(A) [FeCl(CN)4(O2)]4- ; O2 is O22- ; CN- is strong field ligand, so compels for the pairing of electrons.
Fe3+ → 3d5 ;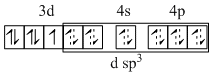
(B) K4[Fe(CN)6] ; CN- is strong field ligand so compels for the pairing of electrons.
Fe2+ → 3d6 :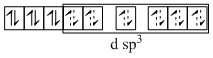
(C) [Co(NH3)6]Cl3 ; NH3 is strong field ligand and 3d6 configuration has higher CFSE compelling for the pairing of electrons.
Co3+ → 3d6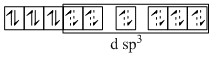
(D) [Fe(CN)5(O2)]5- ; O2 is O22- ; CN- is strong field ligand so compels for the pairing of electrons.
Fe2+ → 3d6 ;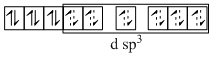
Fe3+ → 3d5 ;
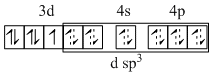
(B) K4[Fe(CN)6] ; CN- is strong field ligand so compels for the pairing of electrons.
Fe2+ → 3d6 :

(C) [Co(NH3)6]Cl3 ; NH3 is strong field ligand and 3d6 configuration has higher CFSE compelling for the pairing of electrons.
Co3+ → 3d6

(D) [Fe(CN)5(O2)]5- ; O2 is O22- ; CN- is strong field ligand so compels for the pairing of electrons.
Fe2+ → 3d6 ;

Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Identify the CORRECT set of details from the following :A. $\left\lbrack Co\left( {NH}_{3} \right)_{6} \right)^{3 +}$ : ...The hypothrtical complex chlorodiaqatriamminecobalt (III)chloride can be represented as...Which of the following complexes is used to be as an anticancer agent?...The correct order of magnetic moments is:...Which of the following will produce a white precipitate upon reacting with AgNO3 ?...