Coordination CompoundHard
Question
Which of the following statements is/are correct ?
Options
A.Ni(CO)4 − Tetrahedral, paramagnetic
B.Ni(CN)4]2- − Square planar, diamagnetic
C.Ni(dmg)2 - Square planar, diamagnetic
D.[NiCl4]2- - Tetrahedral, paramagnetic
Solution
In complex nickel is in zero oxidation state. The CO is strong field ligand and therefore compels for the pairing of electrons. The hybridisation scheme is as shown in figure.
Ni0([Ar] 3d8 4s2)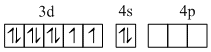
[Ni(CO)4]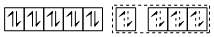 Four pairs of electrons from four CO.
Four pairs of electrons from four CO.
sp3 hybrid orbitals
It is tetrahedral and as all electrons are paired so diamagnetic.
Ni0([Ar] 3d8 4s2)

[Ni(CO)4]
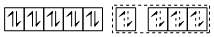 Four pairs of electrons from four CO.
Four pairs of electrons from four CO.sp3 hybrid orbitals
It is tetrahedral and as all electrons are paired so diamagnetic.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Low spin complex is formed by :...Which complex is likely to show optical activity:...Cis-trans isomerism is found in square planar complexes of molecular formula (′a′ and ′b′ are mo...The complexes given below show: and...A compound is made by mixing cobalt (III) nitrite and potassium nitrite solutions in the ratio of 1 : 3. The aqueous sol...