Coordination CompoundHard
Question
What will be the theoretical value of ′spin only′ magnetic moment when Fe(SCN)3 reacts with a solution containing F- ions to yield a colourless complex ?
Options
A.2.83 B.M.
B.3.87 B.M
C.5.92 B.M.
D.1.73 B.M.
Solution
[Fe(SCN)(H2O)5]2+ 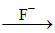 [FeF6]3-, F- being weak ligand Fe3+ -
[FeF6]3-, F- being weak ligand Fe3+ - 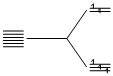 . Therefore, five
. Therefore, five
unpaired electrons and so μ =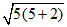 B.M. ≈ 5.92.
B.M. ≈ 5.92.
 . Therefore, five
. Therefore, fiveunpaired electrons and so μ =
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following shows maximum molar conductance ?...In which of the following complexes more than one type of structural isomerism is possible ?...No. of stereoisomer of [Pt(NH3) (py)ClBr] are:...Which one is not an organometallic compound ?...Which one is the most likely structure of CrCl3·6H2O if 1/3 of total chlorine of the compound is precipitated by adding ...