Coordination CompoundHard
Question
[Co(en)3]3+ ion is expected to show ;
Options
A.two optically active isomers: d and l forms
B.d, l and meso forms
C.four optically active isomers: cis, d and l isomers and trans d and l isomers
D.none of these
Solution
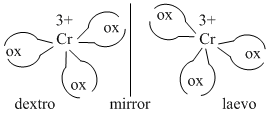
[Co(en)3]3+ ion due to absence of one of the symmetry elements it has two d- and l-optically active forms.

Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
A compound is made by mixing cobalt (III) nitrite and potassium nitrite solutions in the ratio of 1 : 3. The aqueous sol...The EAN of metal atoms in [Fe(CO)2(NO+)2] and Co2(CO)8 respectively are :...A complex [Col6]n+ where L is netural ligand has a magnetic moment μ = 4.5 B. M.Hence...Among the following pairs of ions, the lower oxidation state in aqueous solution is more stable than the other, in...The correct order the wavelength of absorption in the visible region is :...