Coordination CompoundHard
Question
Match Column-I with Column-II and select the correct answer with respect to hybridisation using the codes given below :
Column - I Column - II
(Complex) (Hybridisation)
(I) [Au F4]- (p) dsp2 hybridisation
(II) [Cu(CN)4]3- (q) sp3 hybridisation
(III) [Co(C2O4)3]3- (r) sp3d2 hybridisation
(IV) [Fe(H2O)5NO]2+ (s) d2sp3 hybridisation
Column - I Column - II
(Complex) (Hybridisation)
(I) [Au F4]- (p) dsp2 hybridisation
(II) [Cu(CN)4]3- (q) sp3 hybridisation
(III) [Co(C2O4)3]3- (r) sp3d2 hybridisation
(IV) [Fe(H2O)5NO]2+ (s) d2sp3 hybridisation
Options
A.(I) → q
(II) → p
(III) → r
(IV) → s
(II) → p
(III) → r
(IV) → s
B.(I) → p
(II) → q
(III) → s
(IV) → r
(II) → q
(III) → s
(IV) → r
C.(I) → p
(II) → q
(III) → r
(IV) → s
(II) → q
(III) → r
(IV) → s
D.(I) → q
(II) → p
(III) → s
(IV) → r
(II) → p
(III) → s
(IV) → r
Solution
(I) Au in +3 oxidation state with 5d8 configuration has higher CFSE. So complex has dsp2 hybridisation and is diamagnetic.
(II) Cu is in +1 oxidation state with 3d10 configuration and no (n -1)d orbital is available for dsp2 hybridisaiton, so ns and np orbitals undergo sp3 hybridisation and complex is diamagnetic.
(III) Co is in +3 oxidation state and 3d6 configuration has higher CFSE. So complex is diamagnetic and has d2sp3 hybridisation.
(IV) Fe is in +1 oxidation state and the complex is paramagnetic with three unpaired electrons.
[Fe(H2O)5NO]2+ ;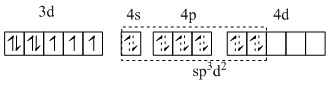
(II) Cu is in +1 oxidation state with 3d10 configuration and no (n -1)d orbital is available for dsp2 hybridisaiton, so ns and np orbitals undergo sp3 hybridisation and complex is diamagnetic.
(III) Co is in +3 oxidation state and 3d6 configuration has higher CFSE. So complex is diamagnetic and has d2sp3 hybridisation.
(IV) Fe is in +1 oxidation state and the complex is paramagnetic with three unpaired electrons.
[Fe(H2O)5NO]2+ ;

Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
50 ml of 0.2 M solution of a compound with empirical formula CoCl3.4NH3 on treatment with excess of AgNO3(aq) yields 1.4...An excess of AgNO3 is added to 100 mL of a 0.01M solution of dichlorotetraaquachromium(III)chloride. The number of moles...The complex, [ Pt (Py) (NH3) BrCl ] will have how many geometrical isomers ?...Some salts although containing two different metallic elements give test for one of them in solution. Such salts are...In which of the following complexes the nickel metal is in highest oxidation state ?...