HydrocarbonsHard
Question
Which one of the following alkenes will react fastest with H2 under catalytic hydrogenation condition ?
Options
A.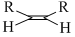

B.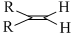

C.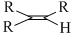

D.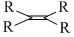

Solution
The rate of hysrogenation of double bond at form. temperature is in the order
R - CH = CH2 > R - CH = CH - R or a ring double bond.
Alkenes of type R2C = CR2 or R2CH = CHR is hydrogenated with difficulty .Thus reactivity order is :
(a) RCH = CH2 (b) R2C = CH2
(1) (2)
(c) R2C = CHR (d) R2C = CR2
(3) (4)
R - CH = CH2 > R - CH = CH - R or a ring double bond.
Alkenes of type R2C = CR2 or R2CH = CHR is hydrogenated with difficulty .Thus reactivity order is :
(a) RCH = CH2 (b) R2C = CH2
(1) (2)
(c) R2C = CHR (d) R2C = CR2
(3) (4)
Create a free account to view solution
View Solution FreeMore Hydrocarbons Questions
The compound formed when 2-butene is treated with hot alk KMnO4 is -...Toluene is aromatisation product of -...Identify Z in the sequence,CH3 − CH2 − CH = CH2...For preparing R − C ≡ CH. From Grignard′s reagent we take -...In the complete combustion of CnH2n+2, the number of oxygen moles required is -...