HydrocarbonsHard
Question
In the complete combustion of CnH2n+2, the number of oxygen moles required is -
Options
A.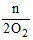
B.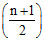 O2
O2
C.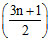 O2
O2
D.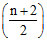 O2
O2
Solution
by balancing the reaction and it is factual also
Create a free account to view solution
View Solution FreeMore Hydrocarbons Questions
Which of the following reactions will result in the formation of a chiral centre in the product -...The major product obtained in the acid-catalysed dimerization of 2-methylpropene is -...Sodium propynide is allowed to react with 2-bromo-2-methylpropane. The major product formed is...Which one of the following alkenes will react fastest with H2 under catalytic hydrogenation condition ?...Alkanes are readily attacked by -...