MetallurgyHard
Question
Which of the following metals cannot be extracted by the carbon reduction process ?
Options
A.Zn
B.Fe
C.Al
D.Sn
Solution
Aluminium is extracted by electrolytic reduction of mixture of molten Al2O3 + Na3AlF6 + CaF2.
Due to very high energy of dissociation of Al2O3, the reduction at such high temperature will give carbide in place of metallic Al according to the following reaction. 2Al2O3 + 6C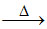 Al4C3 + 3CO2.
Al4C3 + 3CO2.
Due to very high energy of dissociation of Al2O3, the reduction at such high temperature will give carbide in place of metallic Al according to the following reaction. 2Al2O3 + 6C
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
In metallurgical process of aluminium, cryolite is mixed with alumina its molten statebecause it...Van Arkel method of purification of metals involves converting the metal to :...Aluminium is extracted from alumina (Al2O3) by electrolysis of a molten mixture of :...In which of the following pair of metals, both are commercially extracted from their respective ores by self reduction m...In electrolytic refining of lead, electrolyte is consist of :...