MetallurgyHard
Question
In which of the following pair of metals, both are commercially extracted from their respective ores by self reduction method ?
Options
A.Zn, Cu
B.Pb, Cu
C.Sn, Zn
D.Al, Ag
Solution
(i) Extraction of tin (carbon reduction) :
SnO2 + C → SnO + CO↑
(ii) Extraction of zinc (carbon reduction) :
ZnO + C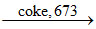 Zn + CO
Zn + CO
(iii) Extraction of lead (self reduction) :
PbS + 2O2 → PbSO4 ; PbS + 3O2 → 2PbO + 2SO2
PbS + 2PbO → 3Pb + SO2 ; PbS + PbSO4 → 2Pb + 2SO2
(iv) Extraction of copper (self reduction) :
2CuFeS2 + 4O2 → Cu2S + 2FeO + 3SO2
Cu2S + FeO + SiO2 → FeSiO3 (fusible slag) + Cu2S (matte)
2Cu2S + 3O2 → 2Cu2O + 2SO2 ; 2Cu2O + Cu2S → 6Cu + SO2
(v) Extraction of aluminium (electrolytic reduction, Hall-Heroult process) :
The purified Al2O3 is mixed with Na3AlF6 (cryolite) or CaF2 (fluorspar) ) which lowers the melting point of the mixture and increases conductivity. The fused matrix is electrolysed.
Cathode : Al3+ (melt) + 3e- Al(l)
Anode : C(s) + O2- (melt) CO(g) + 2e-
C(s) + 2O2- (melt) CO2 (g) + 4e-
(vi) Extraction of gold/silver (leaching and displacement method) :
4Au / Ag (s) + 8CN-(aq) + 2H2O(aq) + O2(g) → 4[Au / Ag (CN)2]-(aq) + 4OH-(aq)
2[Au / Ag (CN)2]-(aq) + Zn(s) → 2Au / Ag (s) + [Zn(CN)4]2- (aq)
SnO2 + C → SnO + CO↑
(ii) Extraction of zinc (carbon reduction) :
ZnO + C
(iii) Extraction of lead (self reduction) :
PbS + 2O2 → PbSO4 ; PbS + 3O2 → 2PbO + 2SO2
PbS + 2PbO → 3Pb + SO2 ; PbS + PbSO4 → 2Pb + 2SO2
(iv) Extraction of copper (self reduction) :
2CuFeS2 + 4O2 → Cu2S + 2FeO + 3SO2
Cu2S + FeO + SiO2 → FeSiO3 (fusible slag) + Cu2S (matte)
2Cu2S + 3O2 → 2Cu2O + 2SO2 ; 2Cu2O + Cu2S → 6Cu + SO2
(v) Extraction of aluminium (electrolytic reduction, Hall-Heroult process) :
The purified Al2O3 is mixed with Na3AlF6 (cryolite) or CaF2 (fluorspar) ) which lowers the melting point of the mixture and increases conductivity. The fused matrix is electrolysed.
Cathode : Al3+ (melt) + 3e- Al(l)
Anode : C(s) + O2- (melt) CO(g) + 2e-
C(s) + 2O2- (melt) CO2 (g) + 4e-
(vi) Extraction of gold/silver (leaching and displacement method) :
4Au / Ag (s) + 8CN-(aq) + 2H2O(aq) + O2(g) → 4[Au / Ag (CN)2]-(aq) + 4OH-(aq)
2[Au / Ag (CN)2]-(aq) + Zn(s) → 2Au / Ag (s) + [Zn(CN)4]2- (aq)
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
In the commercial electrochemical process for aluminium extraction the electrolyte used is...Silver can be separated from lead by :...Refining of silver is done by:...Formation of volatile Ni(CO)4 and then its subsequent decomposition into Ni and CO makes basis of Mond′s process :...Which one of the following is not a method of concentration of ore ?...