General Organic ChemistryHard
Question
Which of the following hydrocarbons has the lowest dipole moment ?
Options
A.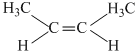
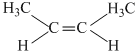
B.CH3 - C ≡ C - CH3
C.CH3 - CH2C ≡ C - CH
D.CH2 = CH - C = CH
Solution
For CH3C = CCH3, μ = 0 because it is symmetrical planar, linear molecule, while others are not.
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
Consider following benzyl alcohol I II III IVCorrect order of their Kb value Is...Give the correct order of decreasing basicity of the following compounds,C6H5NH2 (C6H5)2NH (C6H5)3N C6H11NH2 I II III IV...Which of the following belongs to + I group...In CH3CH2OH, the bond that undergoes heterolytic cleavage most readily is :...Rank the following free radicals in order of decreasing stability : (I) C6H5 HC6H5(II) C6H5 − H − CH = CH2(I...