General Organic ChemistryHard
Question
Rank the following free radicals in order of decreasing stability :
(I) C6H5 HC6H5
(II) C6H5 − H − CH = CH2
(III) CH3 − H − CH3
(IV) C6H5 − H − CH3
(V) CH3CH = CHCH2H2
(VI) CH3 − CH2 −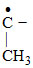 CH3
CH3
(I) C6H5 HC6H5
(II) C6H5 − H − CH = CH2
(III) CH3 − H − CH3
(IV) C6H5 − H − CH3
(V) CH3CH = CHCH2H2
(VI) CH3 − CH2 −
Options
A.I > II > III > IV > V > VI
B.VI > V > IV > III > II > I
C.I > II > IV > VI > III > V
D.I > IV > VI > V > II > III
More General Organic Chemistry Questions
Among the following compounds the one that is most reactive towards electrophilic nitration is:...Arrange pH of the given compounds in decreasing order: (1) Phenol (2) Ethyl alcohol (3) Formic acid (4) Benzoic acid...Y The product Y is...How many optically active stereosiomers are possible for butane -2, 3- diol ?...Increasing of the following for SE I II III IV...