MetallurgyHard
Question
Consider the following statements :
S1 : Extraction of zinc from sphalerite involves roasting followed by carbon reduction.
S2 : In bessemer convertor, along with copper FeSiO3 is also obtained.
S3 : In extraction of lead, CaO prevents the formation of PbSiO3.
S4 : Copper is extracted by hydrometallurgy from low grade ores and scraps.
and arrange in the order of true/false.
S1 : Extraction of zinc from sphalerite involves roasting followed by carbon reduction.
S2 : In bessemer convertor, along with copper FeSiO3 is also obtained.
S3 : In extraction of lead, CaO prevents the formation of PbSiO3.
S4 : Copper is extracted by hydrometallurgy from low grade ores and scraps.
and arrange in the order of true/false.
Options
A.T T T F
B.F T T F
C.T T T T
D.F F F T
Solution
S1 : 2ZnS (sphalerite) + 3O2 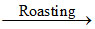 2ZnO + 2SO2 ; ZnO + C
2ZnO + 2SO2 ; ZnO + C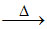 Zn + CO
Zn + CO
S2 : 2CuFeS2 + O2 → Cu2S + 2FeS + SO2
2Cu2S + 3O2 → 2Cu2O + 2SO2 ; 2FeS + 3O2 → 2FeO + 2SO2 ;
FeO + SiO2 → FeSiO3 ; Cu2S + 2Cu2O → 6Cu + SO2
S3 : PbSiO3 + CaO → CaSiO3 + PbO
S4 : 2Cu2O + 4H2SO4 + O2 → 4CuSO4 + 4H2O
Cu2S + 4H2SO4 + 4O2 → 4CuSO4 + 4H2O + 2SO2 ↑
Cu2+ (aq) + H2(g) → Cu(s) + 2H+ (aq)
S2 : 2CuFeS2 + O2 → Cu2S + 2FeS + SO2
2Cu2S + 3O2 → 2Cu2O + 2SO2 ; 2FeS + 3O2 → 2FeO + 2SO2 ;
FeO + SiO2 → FeSiO3 ; Cu2S + 2Cu2O → 6Cu + SO2
S3 : PbSiO3 + CaO → CaSiO3 + PbO
S4 : 2Cu2O + 4H2SO4 + O2 → 4CuSO4 + 4H2O
Cu2S + 4H2SO4 + 4O2 → 4CuSO4 + 4H2O + 2SO2 ↑
Cu2+ (aq) + H2(g) → Cu(s) + 2H+ (aq)
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Calcium silicate slag formed in extraction of iron...In which of the following extration no reducing agent is required ?...Ag2S + NaCN + Zn → AgThis method of extraction of Ag by complex formation and then its displacement is called:...Collectors are the substances which help in attachment of an ore particle to air bubble in froth. A popular collector us...Bauxite is leached with :...