General Organic ChemistryHard
Question
Polarisation of electrons in acrolein may be written as :
Options
A.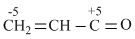
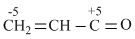
B.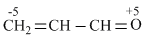
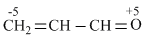
C.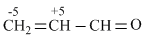
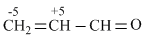
D.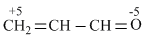
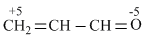
Solution
Because oxygen is more electronegative than carbon, so, that structure is more stable in which partial negative charge is situated at O-atom and partial positive charge on C-atom . This condition is fulled in (d).
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
Consider the following compoundWhich of the above compounds reacts with NaHCO3 giving CO2...Consider the reaction:RCHO + NH2NH2 → RCH = N - NH2 What sort of reaction is it?...Shifting of electron of a multiple bond under the influence of a reagent is called -...The species CH3HCH3 is less stable than -...Rank the following free radicals in order of decreasing stability : (I) C6H5 HC6H5(II) C6H5 − H − CH = CH2(I...