HydrocarbonsHard
Question
Consider the following steps–
(i) CH3 - CH2 - CH3 +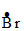
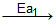 CH3 - CH2 -
CH3 - CH2 - 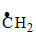 + HBr
+ HBr
(ii) CH3 - CH2 - CH3 +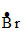
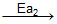 CH3 -
CH3 -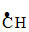 - CH3 + HBr
- CH3 + HBr
Ea1 and Ea2 are activation energies, choose the correct alternative.
(i) CH3 - CH2 - CH3 +
(ii) CH3 - CH2 - CH3 +
Ea1 and Ea2 are activation energies, choose the correct alternative.
Options
A.Ea1 = Ea2
B.Ea1 > Ea2
C.Ea1 < Ea2
D.none of these
Solution
2 degree free radical is more stable
Create a free account to view solution
View Solution FreeMore Hydrocarbons Questions
The major product formed in the reactions is...Which can yield acetylene in single step...Consider the following reaction sequence. CH ≡ CH A B The products(A) and (B) are, respectively,...A hydrocarbon, ′X′(C3H4) decolourise bromine in carbon tetrachloride forming ′Y′ . ′X̸...The compound which has one isopropyl group is :...