MetallurgyHard
Question
The extraction of zinc from zinc blende involves :
Options
A.the electrolytic reduction.
B.the roasting followed by reduction with carbon.
C.the calcination followed by reduction with another metal.
D.the roasting at molten temperature.
Solution
2ZnS + 3O2 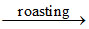 2ZnO + 2SO2
2ZnO + 2SO2
ZnO + C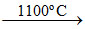 Zn + CO.
Zn + CO.
Therefore, (B) option is correct.
ZnO + C
Therefore, (B) option is correct.
Create a free account to view solution
View Solution Free