MetallurgyHard
Question
Extraction of silver from Ag2S by the use of sodium cyanide is an example of :
Options
A.roasting
B.hydrometallurgy
C.electrometallurgy
D.smelting
Solution
Ag2S (concentrated ore) + 2NaCN (aq) 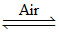 2AgCN (s) + Na2S (aq).
2AgCN (s) + Na2S (aq).
4Na2S (aq) + 5O2 (g) + 2H2O (l) → 2Na2SO4 (aq) + 4NaOH (aq) + 2S (s)
AgCN (s) + NaCN (aq) → Na[Ag(CN)2] (soluble complex)
2Na[Ag(CN)2] (aq) + Zn (dust) → 2Ag (s) + Na2[Zn(CN)4] (aq). Hence these reactions suggest that extraction of silver from Ag2S by the use of sodium cyanide is an example of hydrometallurgy.
4Na2S (aq) + 5O2 (g) + 2H2O (l) → 2Na2SO4 (aq) + 4NaOH (aq) + 2S (s)
AgCN (s) + NaCN (aq) → Na[Ag(CN)2] (soluble complex)
2Na[Ag(CN)2] (aq) + Zn (dust) → 2Ag (s) + Na2[Zn(CN)4] (aq). Hence these reactions suggest that extraction of silver from Ag2S by the use of sodium cyanide is an example of hydrometallurgy.
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Calcination is the process of heating the ore:...Which one of the following is not a method of concentration of ore ?...Aluminium is extracted from alumina (Al2O3) by electrolysis of a molten mixture of :...Which of the following reduction reactions are actually employed in commercial extraction of metals?...Cryolite is :...