MetallurgyHard
Question
In the extraction of Cu the reaction takes place in Bessemer converter is :
Options
A.2Cu2O + Cu2S → 6Cu + SO2.
B.2CuFeS2 + O2 → Cu2S + FeS + SO2
C.2Cu2S + 3O2 → 2Cu2O + 2SO2
D.2FeS + 3O2 → 2FeO + 2SO2
Solution
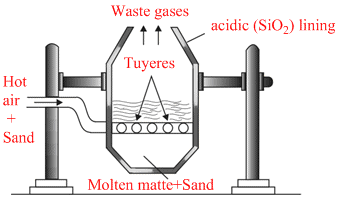
Bessemer convertor
Reactions involved :
2FeS + 3O2 → 2FeO + 2SO2 ; FeO + SiO2 → FeSiO3
2Cu2S + 3O2 → 2Cu2O + 2SO2 ; 2Cu2O + Cu2S → 6Cu + SO2 (self reduction)
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Gravity separation method is based upon :...It is noticed that ${Pb}^{2 +}$ is more stable than ${Pb}^{2 +}$ but ${Sn}^{2 +}$ is less stable than ${Sn}^{4 +}$. Obse...Ag2S + NaCN + Zn → AgThis method of extraction of Ag by complex formation and then its displacement is called:...Which of th following statement is correct about ionization potential ?...Out of Cu2S, HgS, Ag2S, PbS and ZnS, roasting will convert the minerals into metal in case of :...