Photoelectric EffectHard
Question
The energy of a hydrogen atom in the ground state is - 13.6 eV. The energy of He+ ion in the first excited state will be
Options
A.- 13.6 eV
B.- 27.2 eV
C.- 54.4 eV
D.- 6.8 eV
Solution
Energy of an hydrogen like atom like He+ in an nth orbit is given by
En =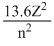 eV
eV
For hydrogen atom, Z = 1
∴ En = -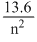 eV
eV
For ground state, n = 1
∴ En = -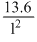 eV = - 13.6 eV
eV = - 13.6 eV
For He+ ion Z = 0
En = -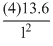 eV
eV
For excited state, n = 2
∴ E2 = -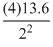 eV = - 13.6 eV
eV = - 13.6 eV
Hence, the energy in He+ ion in first excited state is the same that of energy of the hydrogen atom in ground state i.e., -13.6 eV.
En =
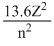 eV
eVFor hydrogen atom, Z = 1
∴ En = -
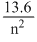 eV
eVFor ground state, n = 1
∴ En = -
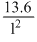 eV = - 13.6 eV
eV = - 13.6 eVFor He+ ion Z = 0
En = -
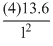 eV
eVFor excited state, n = 2
∴ E2 = -
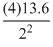 eV = - 13.6 eV
eV = - 13.6 eVHence, the energy in He+ ion in first excited state is the same that of energy of the hydrogen atom in ground state i.e., -13.6 eV.
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
Photoelectrons are emitted by a metal surface only, when :...Hydrogen atom in ground state is excited by a monochromatic radiation of λ = 975. Number ofspectral lines in the re...According to Mosley′s law, the frequency of a characteristic spectral line in X-ray spectrum varies as :-...A point source of light is used in a photoelectric effect. If the source is moved toward the emitting metal, the stoppin...Energy of a photon can not represented by...