AIPMT | 2014Photoelectric EffectHard
Question
Hydrogen atom in ground state is excited by a monochromatic radiation of λ = 975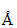 . Number ofspectral lines in the resulting spectrum emitted will be
. Number ofspectral lines in the resulting spectrum emitted will be
Options
A.3
B.2
C.6
D.10
Solution
Energy incident = 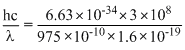 eV
eV
= 12.75 eV
The Hydrogen atom will be excited to n = 4
Number of spectral lines =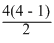 = 6
= 6
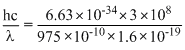 eV
eV= 12.75 eV
The Hydrogen atom will be excited to n = 4
Number of spectral lines =
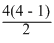 = 6
= 6Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
When light is incident on surface, photo electrons are emitted. For photoelectrons :...Let n1 be the frequency of the series limited of the Lyman series, ν2 be the frequency of the first line of the Lym...The phenomenon of photoelectric effect was explained by :...When light of wavelength lesser than 6000 is incident on a metal, electrons are emitted. The approximate work-function o...For constant hydraulic streess on an object, the fractional change in the object′svolume (ᐃV/ V) and its bul...