Analytical ChemistryHard
Question
A metal M and its compound can give the following observable changes in a sequence of reactions, M 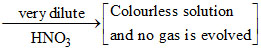

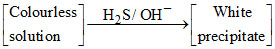 The metal M can be :
The metal M can be :
Options
A.Mg
B.Pb
C.Zn
D.Sn
Solution
4Zn + 10H+ + NO3- → 4Zn2+ + NH4+ + 3H2O.
Zn2+ + 2OH- → Zn(OH)2↓ (white) ; Zn(OH)2 + 2OH- → [Zn(OH)4]2-.
[Zn(OH)4]2- + H2S → ZnS ↓ (white) + 2OH- + 2H2O.
Zn2+ + 2OH- → Zn(OH)2↓ (white) ; Zn(OH)2 + 2OH- → [Zn(OH)4]2-.
[Zn(OH)4]2- + H2S → ZnS ↓ (white) + 2OH- + 2H2O.
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
When NH4Cl is added to a solution of NH4OH :...A solution of colourless salt H on boiling with excess NaOH produces a non-flammable gas. The gas evolution ceases after...27 g of aluminium react completely with how many grams of oxygen ?...An aqueous solution contains both Al3+ & Zn2+.To this solution NH4OH is added in excess....Select the correct statement(s)....