Analytical ChemistryHard
Question
Which of the following metal salts gives a red and opaque borax bead in the reducing flame (in cold) ?
Options
A.Ni
B.Fe
C.Cu
D.Mn
Solution
Na2B4O7 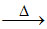 2NaBO2 + B2O3
2NaBO2 + B2O3
CuO + B2O3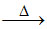 Cu(BO2)2
Cu(BO2)2
2Cu(BO2)2 + 4NaBO2 + 2C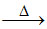 2Cu (red and opaque) + 2Na2B4O7 + 2CO ↑
2Cu (red and opaque) + 2Na2B4O7 + 2CO ↑
CuO + B2O3
2Cu(BO2)2 + 4NaBO2 + 2C
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
Select the correct statement with respect to Fe3+ ions....An aqueous solution of salt containing an acidic radical X- reacts with sodium hypochlorite in neutral medium. The gas e...CrO42- + H+ + H2O2 X + H2OIdentify the correct statement with respect to X....White precipitate of AgCl turns to greyish or black when :...Which of the following reagents gives a yellow precipitate with a hot faintly acidic solution of Bi3+ ions ?...