Analytical ChemistryHard
Question
To a solution of a substance, gradual addition of ammonium hydroxide results in a brownish black precipitate which does not dissolve in excess of NH4OH. However, when KI (not in excess) is added to the original solution, a green precipitate is formed. The solution contained :
Options
A.Lead salt
B.Silver salt
C.Mercurous salt
D.Copper salt.
Solution
Hg2Cl2 + 2NH3 → 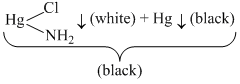 + NH4Cl
+ NH4Cl
Hg22+ + 2I- → Hg2I2 ↓ (green)
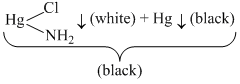 + NH4Cl
+ NH4ClHg22+ + 2I- → Hg2I2 ↓ (green)
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
An aqueous solution contains both Al3+ & Zn2+.To this solution NH4OH is added in excess....Which of the following will not give positive chromyl chloride test ?...The compound used for gravimetric estimation of copper (II) is :...Nessler′s reagent is :...Consider the reactions shown below ; Which of the following statement (s) is/are correct ?...