Analytical ChemistryHard
Question
Consider the reactions shown below ;
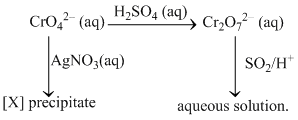
Which of the following statement (s) is/are correct ?
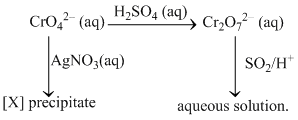
Which of the following statement (s) is/are correct ?
Options
A.[X] is a yellow coloured precipitate.
B.[X] is soluble in ammonia solution.
C.[Y] gives green coloured solution with excess of sodium hydroxide solution.
D.The conversion of Cr2O72- to [Y] is an redox reaction.
Solution
(A) Ag+ 2CrO42- → Ag2CrO4 ↓ (red) [X].
(B) Ag2CrO4 ↓ + 4NH3 → 2[Ag(NH3)2]+ + CrO42-.
(C) Cr3+ + 3OH- → Cr(OH)3 ↓ (green).
Cr(OH)3 + OH- → [Cr(OH)4]- (green coloured soluble complex).
(D) 3SO2+4 + Cr2+7O72- + 2H+ → 2Cr3+[Y] + 3S+6O42- + H2O.
(B) Ag2CrO4 ↓ + 4NH3 → 2[Ag(NH3)2]+ + CrO42-.
(C) Cr3+ + 3OH- → Cr(OH)3 ↓ (green).
Cr(OH)3 + OH- → [Cr(OH)4]- (green coloured soluble complex).
(D) 3SO2+4 + Cr2+7O72- + 2H+ → 2Cr3+[Y] + 3S+6O42- + H2O.
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
A metal chloride original solution (i.e. O.S) on mixing with K2CrO4 solution gives a yellow precipitate soluble in aqueo...Which of the following statement(s) is/are correct with respect to bromide ions ?...Ammonia/ammonium ion gives yellow precipitate with :...In the fourth group, white precipitate of Mn(OH)2 on heating with PbO2 and concentrated HNO3 gives red-violet (purple) c...An aqueous solution of salt gives white precipitate with AgNO3 solution as well as with dilute H2SO4. It may be...