Analytical ChemistryHard
Question
Na2 SO4 + BaCl2 → White precipitate 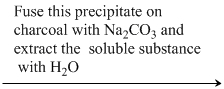 Aqueous solution.
Aqueous solution.  Gas ′G′ is evolved
Gas ′G′ is evolved
The gas ′G′ will show which of the following property ?
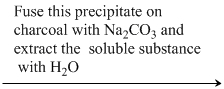 Aqueous solution.
Aqueous solution.  Gas ′G′ is evolved
Gas ′G′ is evolvedThe gas ′G′ will show which of the following property ?
Options
A.It turns lead acetate filter paper black.
B.It turns acidified K2Cr2O7 filter paper green.
C.It produces purple colouration on filter paper moistened with sodium nitroprusside already made alkaline with sodium hydroxide.
D.All of these
Solution
(A) Pb2+ + H2S → PbS ↓ (black) + 2H+.
(B) Cr2O72- + 3H2S + 8H+ → 2Cr3+ (green colour solution) + 3S↓ + 7H2O.
(C) S2- + [Fe(CN)5NO]2- → [Fe(CN)5NOS]4- (purple colouration).
Reactions : Na2SO4 + BaCl2 → BaSO4 ↓ + NaCl.
BaSO4 + 4C + Na2CO3 Na2S + BaCO3 ↓ + 4CO ↑.
Na2S + BaCO3 ↓ + 4CO ↑.
Na2S + H2SO4 → Na2SO4 + H2S.
(B) Cr2O72- + 3H2S + 8H+ → 2Cr3+ (green colour solution) + 3S↓ + 7H2O.
(C) S2- + [Fe(CN)5NO]2- → [Fe(CN)5NOS]4- (purple colouration).
Reactions : Na2SO4 + BaCl2 → BaSO4 ↓ + NaCl.
BaSO4 + 4C + Na2CO3
Na2S + H2SO4 → Na2SO4 + H2S.
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
A solution containing SCN- ions can be used to test one or more out of : Fe3+, Co2+ , Cu2+, Ag+ and Hg2+....Which of the statements is not true?...An aqueous solution of a substance gives a white precipitate on treatment with dilute hydrochloric acid, which dissolves...Which of the following statement(s) is/are correct with respect to bromide ions ?...Which one of the following can be used in place of NH4Cl for the identification of the third group radicals?...