Analytical ChemistryHard
Question
A solution containing SCN- ions can be used to test one or more out of : Fe3+, Co2+ , Cu2+, Ag+ and Hg2+.
Options
A.Fe3+ and Co2+ only
B.Co2+, Cu2+, Ag+ and Hg2+
C.Fe3+ , Cu2+, Co2+ and Hg2+
D.All
Solution
CoCl2 + 4NH4SCN 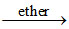 (NH4)2 [Co(SCN)4] (blue colour in ethereal layer) + 2NH4Cl.
(NH4)2 [Co(SCN)4] (blue colour in ethereal layer) + 2NH4Cl.
Cu2+ + 3SCN- → CuSCN↓ (white) + (SCN)2.
FeCl3 + 3NH4SCN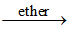 Fe(SCN)3 (blood red colour) + 3NH4Cl.
Fe(SCN)3 (blood red colour) + 3NH4Cl.
Ag+ + SCN- → AgSCN ↓ (white).
Co2+ + 4SCN- + Hg2+ → Co[Hg(SCN)4] ↓ (deep blue).
Cu2+ + 3SCN- → CuSCN↓ (white) + (SCN)2.
FeCl3 + 3NH4SCN
Ag+ + SCN- → AgSCN ↓ (white).
Co2+ + 4SCN- + Hg2+ → Co[Hg(SCN)4] ↓ (deep blue).
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
A solution of colourless salt H on boiling with excess NaOH produces a non-flammable gas. The gas evolution ceases after...Consider the following statements, S1 : sulphide does give yellow precipitate with Cd(NO3)2 in aqueous solution. S2 : aq...Which one of the following cations will give a green coloured ash when a piece of filter paper dipped in a solution cont...A white crystalline substance dissolves in water. On passing H2S in this solution, a black precipitate is obtained. The ...Which of the following cation does not give red colour precipitate/solution with dimethylglyoxime (DMG) in alkaline solu...