Current Electricity and Electrical InstrumentHard
Question
In producing chlorine by electrolysis 100 kW power at 125 V is being consumed. How much chlorine per minute is liberated (E.C.E. of chlorine is 0.367 × 10-6 kg/C)
Options
A.1.76 × 10-3 kg
B.9.67 × 10-3 kg
C.17.61 × 10-3 kg
D.3.67 × 10-3 kg
Solution
Current, I = 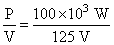 = 800 A
= 800 A
According to the Faraday′s first law electrolysis Mass of clorine liberated, m = zIt.
m = (0.367 × 10-6 kg/C)(800A) (60 s)
m =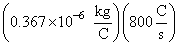 (60s)
(60s)
= 17.61 × 10-3 kg
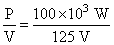 = 800 A
= 800 AAccording to the Faraday′s first law electrolysis Mass of clorine liberated, m = zIt.
m = (0.367 × 10-6 kg/C)(800A) (60 s)
m =
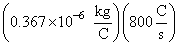 (60s)
(60s)= 17.61 × 10-3 kg
Create a free account to view solution
View Solution FreeTopic: Current Electricity and Electrical Instrument·Practice all Current Electricity and Electrical Instrument questions
More Current Electricity and Electrical Instrument Questions
A wire has resistance of 24Ω is bent in the following shape. The effective resistance between A and B is -...A wire has a non-uniform cross-section as shown in figure. A steady current flows through it. The drift speed of electro...A small coil lies inside a larger coil. The two coils are horizontal, concentric and carry currents in the same directio...In a metre bridge experiment null point is obtained at 20 cm from one end of the wire when resistance X is balanced agai...A wire of uniform resistance $\lambda\Omega/m$ is bent into a circle of radius $r$ and another piece of wire with length...