Analytical ChemistryHard
Question
A coloured solution of an inorganic salt reacts with potassium thiocyanate to give first a black precipitate, which slowly turns white. The salt solution also gives black precipitate with H2S gas in slightly acidic medium. The black precipitate dissolves in potassium cyanide forming a colourless solution. The basic radical present in the inorganic salt is :
Options
A.Bi3+
B.Cu2+
C.Hg2+
D.None
Solution
Cu2+ + 2SCN- → Cu(SCN)2↓ (black)
2Cu(SCN)2 → 2CuSCN↓ (white) + (SCN)2
Cu2+ + H2S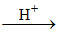 CuS↓ (black) + 2H+
CuS↓ (black) + 2H+
2CuS↓ + 8CN- → 2[Cu(CN)4]3- + S22-
2Cu(SCN)2 → 2CuSCN↓ (white) + (SCN)2
Cu2+ + H2S
2CuS↓ + 8CN- → 2[Cu(CN)4]3- + S22-
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
Potassium chromate solution is added to an aqueous solution of a metal chloride. The yellow precipitate thus obtained is...When a mixture of solid NaCl and solid K2Cr2O7 is heated with concentrated H2SO4, deep red vapours are obtained. This is...The correct option(s) to distinguish nitrate salts of Mn2+ and Cu2+ taken separately is (are) :-...Which of the following reagents gives a yellow precipitate with a hot faintly acidic solution of Bi3+ ions ?...Compound (X) when heated with KOH solution it produce colorless poisonous gas (Y) having rotten fish smell and solution ...