s-Block ElementsHard
Question
Potassium carbonate is not prepared by :
Options
A.Solvay process
B.Le-Blanc process
C.Prechts process
D.none of these.
Solution
(A) KHCO3 obtained as intermediate is water soluble. So it cannot be separated for obtaing K2CO3 by calcination in Solvay process.
(B) Le-Blanc process :
KCl + H2SO4 → KHSO4 + HCl ; KHSO4 + KCl → K2SO4 + HCl
K2SO4 + CaCO3 + 2C → K2CO3 + CaS + 2CO2
(C) Prechts process :
(a) 2KCl + 3(MgCO3. 3H2O) + CO2 → 2(Mg CO3. KHCO3. 4H2O) + MgCl2
(b) 2(MgCO3. KHCO3. 4H2O)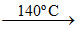 2Mg CO3 ↓ + K2CO3 + 9H2O + CO2.
2Mg CO3 ↓ + K2CO3 + 9H2O + CO2.
(B) Le-Blanc process :
KCl + H2SO4 → KHSO4 + HCl ; KHSO4 + KCl → K2SO4 + HCl
K2SO4 + CaCO3 + 2C → K2CO3 + CaS + 2CO2
(C) Prechts process :
(a) 2KCl + 3(MgCO3. 3H2O) + CO2 → 2(Mg CO3. KHCO3. 4H2O) + MgCl2
(b) 2(MgCO3. KHCO3. 4H2O)
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Among the following components of cement which is present in highest amount ?...Which of the following metal carbonate is/are thermally stable ?...Sodium carbonate can be manufactured by Solvay′s process but potassium carbonate cannot be prepared because:...Chemical (A) is used for water softening to remove temporary hardness. A reacts with Na2 CO3 to generate caustic soda. W...A colourless solid (X) on heating evolved CO2 and also gave a white residue, soluble in water. Residue also gave CO2 whe...