s-Block ElementsHard
Question
A colourless solid (X) on heating evolved CO2 and also gave a white residue, soluble in water. Residue also gave CO2 when treated with dilute acid. (X) is :
Options
A.K2CO3
B.CaCO3
C.KHCO3
D.Na2CO3
Solution
KHCO3 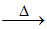 K2CO3 (soluble in water) + CO2 + H2O.
K2CO3 (soluble in water) + CO2 + H2O.
K2CO3 + H2SO4 → K2SO4 + CO2 + H2O.
K2CO3 + H2SO4 → K2SO4 + CO2 + H2O.
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Alkali metals possess metallic lustre when freshly cut because :...Which of the following has lowest thermal stability ?...On commercial scale, sodium hydroxide is prepared by :...Gypsum on heating at a temperature of 393 K yields :...Be and Al have following resemblance due to diagonal relationship :...