s-Block ElementsHard
Question
The compressibility factor for a real gas at highpressure is :-
Options
A.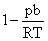
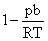
B.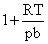
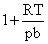
C.1
D.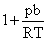
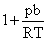
Solution
∴ For real gas, according to Vander wall′sequation
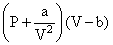 = RT (For 1 mole gas)
= RT (For 1 mole gas)
At high pressure
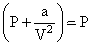
∴ P(V - b) = RT
PV = RT + Pb
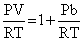
∴ Compressibility factor for a real gas
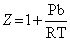
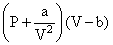 = RT (For 1 mole gas)
= RT (For 1 mole gas)At high pressure
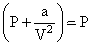
∴ P(V - b) = RT
PV = RT + Pb
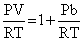
∴ Compressibility factor for a real gas
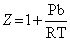
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Sodium carbonate is prepared by:...Peroxide ion is present in :...The deep brown colour produced when iodine is dissolved in a solution of potassium iodide is caused by the presence of:...In which of the following process, Black ash (Na2CO3 + CaS) is obtained ?...The molecular formula of plaster of pairs is :...