s-Block ElementsHard
Question
In which of the following process, Black ash (Na2CO3 + CaS) is obtained ?
Options
A.Le-Blanc process
B.Prechts process
C.Hargreaves process
D.Gossage process
Solution
(A) It is used for the manufacture of Na2CO3 from NaCl
NaCl + H2SO4 (conc.)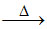 NaHSO4 + HCl
NaHSO4 + HCl
NaHSO4 + NaCl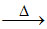 Na2SO4 + HCl
Na2SO4 + HCl
Na2SO4 + CaCO3 + 4 C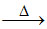
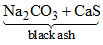 + 4CO
+ 4CO
NaCl + H2SO4 (conc.)
NaHSO4 + NaCl
Na2SO4 + CaCO3 + 4 C
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Lithopone is a mixture of...Property(ies) of all the alkali metals that increase(s) with their atomic number is (are) :...Lithium is stronger reducing agent than sodium due to :...Based on lattice energy and other considerations which one of the following alkali metal chlorides is expected to have t...Acidified solutions of sodium thiosulphate are unstable because :...