s-Block ElementsHard
Question
Consider the following statements,
S1 : KO2 is CO and CO2 abrosber, and O2 emitter.
S2 : CaCl2. 6 H2O is widely used for melting ice on roads, particulary in very cold countries.
S3 : The process of setting of plaster of paris is exothermic.
S4 : MgCl2.6 H2O on strong heating yields anhydrous MgCl2.
and arrange in the order of true/false.
S1 : KO2 is CO and CO2 abrosber, and O2 emitter.
S2 : CaCl2. 6 H2O is widely used for melting ice on roads, particulary in very cold countries.
S3 : The process of setting of plaster of paris is exothermic.
S4 : MgCl2.6 H2O on strong heating yields anhydrous MgCl2.
and arrange in the order of true/false.
Options
A.T T T T
B.F T T T F
C.T T T F
D.T F F T
Solution
S1 : 2 KO2 + CO2 → K2CO3 + 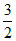 O2
O2
S2 : Because a 30% mixture of CaCl2 / H2O freezes at - 55oC as compared to NaCl / H2O at - 18oC. As such CaCl2 melts down the ice by decreasing the melting point.
S3 : 2 CaSO4. H2O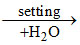 CaSO4 . 2 H2O. (Heat is evolved as a result of the formation of another hydrate.)
CaSO4 . 2 H2O. (Heat is evolved as a result of the formation of another hydrate.)
S4 : MgCl2 . 6 H2O → MgO + 2 HCl + 5 H2O.
S2 : Because a 30% mixture of CaCl2 / H2O freezes at - 55oC as compared to NaCl / H2O at - 18oC. As such CaCl2 melts down the ice by decreasing the melting point.
S3 : 2 CaSO4. H2O
S4 : MgCl2 . 6 H2O → MgO + 2 HCl + 5 H2O.
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Match the compounds /metal in (X) with their uses in (Y) : X YA. liquid sodium metal I breathing apparatus in submarineB...The commercial salt is purified by passing...The molecular formula of plaster of pairs is :...At higher temperature sodium metal reacts with alumina to give a sodium compound ′X′. ′X′ is dis...A compound X on heating gives a colourless gas. The residue is dissolved in water to obtained Y. Excess CO2 is bubbled t...