s-Block ElementsHard
Question
Consider the following statements ;
S1 : Ozone reacts with dry KOH forming a deep red/orange coloured compound and liberating oxygen gas at low temperature.
S2 : KO2 is better oxidant than K2O2 both in aqueous and acidic mediums.
S3 : KOH is better absorber of CO2 than NaOH.
S4 : Quick lime on heating with carbon in an electric furnace forms CaCO3.
and arrange in the order of true/false.
S1 : Ozone reacts with dry KOH forming a deep red/orange coloured compound and liberating oxygen gas at low temperature.
S2 : KO2 is better oxidant than K2O2 both in aqueous and acidic mediums.
S3 : KOH is better absorber of CO2 than NaOH.
S4 : Quick lime on heating with carbon in an electric furnace forms CaCO3.
and arrange in the order of true/false.
Options
A.T T T F
B.T T T T
C.F F T F
D.T F T F
Solution
S1 : KOH + O3 → KO3 (deep red/orange) + O2 + H2O.
S2 : 2 KO2 + 2 H2O → KOH + H2O2 + O2 . ; 2 KO2 + H2SO4 → K2SO4 + H2O2 + O2.
gives both O2 and H2O2, where as K2O2 gives only H2O2
S3 : Because K2CO3 is more soluble in water and does not separate out.
S4 : CaO + 2C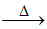 CaC2 + CO.
CaC2 + CO.
S2 : 2 KO2 + 2 H2O → KOH + H2O2 + O2 . ; 2 KO2 + H2SO4 → K2SO4 + H2O2 + O2.
gives both O2 and H2O2, where as K2O2 gives only H2O2
S3 : Because K2CO3 is more soluble in water and does not separate out.
S4 : CaO + 2C
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Alkali metals are not characterised by :...The substance not likely to contain CaCO3 is...Na2CO3 reacts with SO2 in alkaline medium to form:...Which of the following gives sodium hydroxide along with hydrogen gas on reaction with water ?...Match the compounds /metal in (X) with their uses in (Y) : X YA. liquid sodium metal I breathing apparatus in submarineB...