s-Block ElementsHard
Question
Which of the following statement(s) is/are correct ?
Options
A.Pure sodium oxide is obtained by heating the mixture of sodium azide and sodium nitrite.
B.Glauber′s salt effloresces in moist air.
C.Potassium superoxide on heating in an evacuated and sealed tube yields sodium thiosulphate.
D.Gypsum dissovle in ammonium sulphate solution.
Solution
(A) 3NaN3 + NaNO2 → 2Na2O + 5N2.
(B) False → Glauber′s salt (Na2SO4.10H2O) effloresces in dry air.
(C) 2 KO2 + S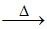 K2SO4.
K2SO4.
(D) Form soluble double sulphate, (NH4)2SO4.CaSO4.H2O (soluble complex).
(B) False → Glauber′s salt (Na2SO4.10H2O) effloresces in dry air.
(C) 2 KO2 + S
(D) Form soluble double sulphate, (NH4)2SO4.CaSO4.H2O (soluble complex).
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Drying agent which react with CO2 and removes water vapours from ammonia is :...When NaNO3 is treated with Na :...Consider the following statements ;S1 : Among alkali metal, halides lithium iodide is the most covalent in nature. S2 : ...Calcium cyanamide on reaction with steam under pressure gives ammonia and -------....Which of the following is different from other three oxides ?...