s-Block ElementsHard
Question
Freshly prepared pure dilute solution of sodium in liquid ammonia :
Options
A.shows copper - bronze colour.
B.occupy larger volume than that from the sum of the volumes of Na and NH3 (l).
C.reduces the GeH4 to GeH2-.
D.produces sodium amide and hydrogen gas with rusty iron wire.
Solution
(A) The alkali metals dissolve in liquid ammonia giving blue solutions.
M + (x + y) NH3 → [M(NH3)x]+ + [e(NH3)y]-
The blue colour, corresponding to a broad absorption band near 1500 nm that tails into the visible range, is attributed to the solvated electron.
(B) According to the cavity concept, they occupy far greater volume than that expected from the sum of the volumes of metal and solvent.
(C) The solution of metals in liquid ammonia act as powerful reducing agents (it even reduces the aromatic ring)
GeH4 + e- → GeH2- +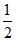 H2.
H2.
(D) In the presence of impurities or transition metals like Fe ; MNH2 and H2 gas are formed.
M + NH3 → MNH2 +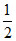 H2
H2
M + (x + y) NH3 → [M(NH3)x]+ + [e(NH3)y]-
The blue colour, corresponding to a broad absorption band near 1500 nm that tails into the visible range, is attributed to the solvated electron.
(B) According to the cavity concept, they occupy far greater volume than that expected from the sum of the volumes of metal and solvent.
(C) The solution of metals in liquid ammonia act as powerful reducing agents (it even reduces the aromatic ring)
GeH4 + e- → GeH2- +
(D) In the presence of impurities or transition metals like Fe ; MNH2 and H2 gas are formed.
M + NH3 → MNH2 +
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Which of the following alkaline earth metal sulphates has hydration enthalpy higher than the lattice enthalpy ?...Dilute solutions of alkali metals in liquid ammonia :...Setting of plaster of paris involves :...Which one of the alkali metals, forms only, the normal oxide, M2O on heating in air ?...Sodium is heated in excess of air, free from CO2 at 350oC to form X. X absorbs CO2 and form Na2CO3 and Y. ′X′...