s-Block ElementsHard
Question
Sodium is heated in excess of air, free from CO2 at 350oC to form X. X absorbs CO2 and form Na2CO3 and Y. ′X′ and ′Y′ are respectively :
Options
A.Na2O and O2
B.Na2O2 and O2
C.NaO2 and O2
D.Na2O2 and O3
Solution
2 Na + O2(air) 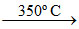 Na2O2 (X)
Na2O2 (X)
2 CO2 + 2 Na2O2 → 2 Na2CO3 + O2 (Y)
Note : Na2O2 + O2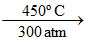 2 NaO2 ; Na + O2 (limited amount)
2 NaO2 ; Na + O2 (limited amount) 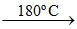 Na2O
Na2O
2 CO2 + 2 Na2O2 → 2 Na2CO3 + O2 (Y)
Note : Na2O2 + O2
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Largest difference radii is found in case of the pair...The first ionisation potential (eV) of Be and B respectively are :...Which of the following has the highest melting point?...Select correct statement(s) :...Which of the alkaline earth metal halides given below is essentially covalent in nature ?...