s-Block ElementsHard
Question
CaCl2 + H2SO4 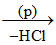 CaSO4.2H2O
CaSO4.2H2O 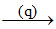 (r)
(r) 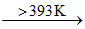 (s)
(s)
Which of the following option describes, the products, reactants and the reaction conditions.
Which of the following option describes, the products, reactants and the reaction conditions.
Options
A.(p) Crystallisation (q) Heat at 393 K (r) 2 CaSO4.H2O (s) CaSO4
B.(p) Crystallisation (q) Heat at 393 K at high pressure (r) 2 CaSO4.H2O (s) CaSO4
C.(p) Higher temperature (q) Cool (r) CaSO4.H2O (s) CaSO4
D.(p) Higher pressure (q) Heat at 393 K (r) CaSO4 (s) CaSO3
Solution
CaCl2 + H2SO4 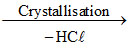 CaSO4.2H2O
CaSO4.2H2O 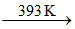 2 CaSO4.H2O
2 CaSO4.H2O 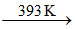 CaSO4.
CaSO4.
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Which one of the following electrolyte is used in Down′s process of extracting sodium metal ?...Low solubility of CsI in water is due to :...Which of the following can not decompose on heating to give CO2?...Going down in II A group, following properties decrease :...Select the correct statement(s) with respect to sodium hydroxide....