s-Block ElementsHard
Question
Which of the following can not decompose on heating to give CO2?
Options
A.Li2CO3
B.Na2CO3
C.KHCO3
D.BaCO3
Solution
Alkali metal carbonates except Li2CO3 are stable towards heat because they are most basic in nature and basic character increases down the group and thermal stability increases down the group.
Bigger HCO3- anion is polarised by smaller Li+ and thus readily decomposes to give CO2 gas.
Li2CO3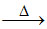 Li2O + CO2
Li2O + CO2
Bigger HCO3- anion is polarised by smaller Li+ and thus readily decomposes to give CO2 gas.
Li2CO3
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Solution of K2O in water is basic, because it contains a significant concentration of :...The metallic lustre exhibited by sodium is explained by :...Hydrated sodium aluminium silicate, the zeolite, is treated with hard water, the sodium ions are exchanged with :...Thermal stability of MCO3 is in order :...The compound(s) formed upon combustion of sodium metal in excess air is(are)...