s-Block ElementsHard
Question
What happens when sodium metal is heated to a temperature 350oC in excess of dry air containing carbon dioxide gas ?
Options
A.Na2O2 is formed.
B.Na2O is formed.
C.First Na2O2 is formed which then converts in to Na2CO3.
D.First NaO2 is formed which then converts into Na2CO3.
Solution
2Na + O2 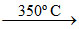 Na2O2 ; 2Na2O2 + 2CO2 → 2Na2CO3 + O2.
Na2O2 ; 2Na2O2 + 2CO2 → 2Na2CO3 + O2.
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Which of the following statement (s) is/are correct with respect to alkali metal bicarbonates ?...Which of the following gives sodium hydroxide along with hydrogen gas on reaction with water ?...Alkali metals possess metallic lustre when freshly cut because :...Which is/are true statements ?...Sodium has, as compared to potassium :...